Botany online 1996-2004. No further update, only historical document of botanical science!
A nucleotide consists of a base (a heterocyclic nitrogen-containing component), a sugar (ribose or desoxyribose) and one or more phosphate residues.
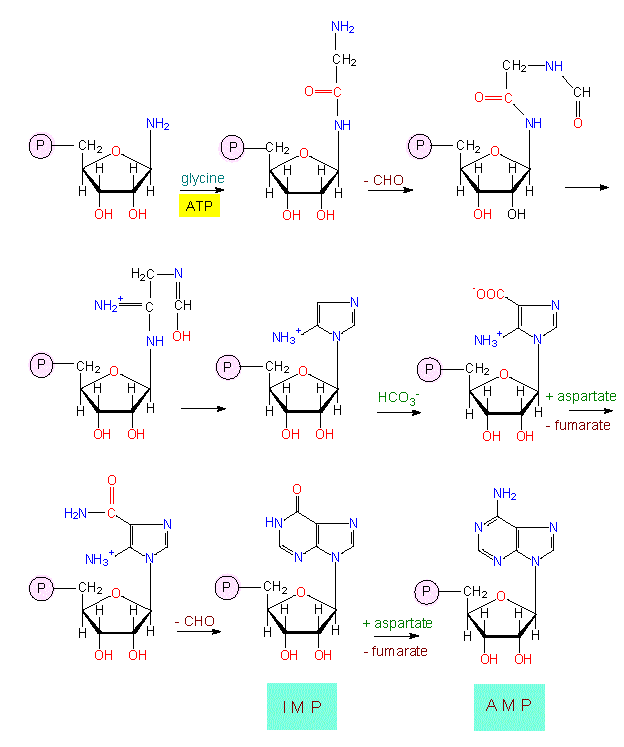
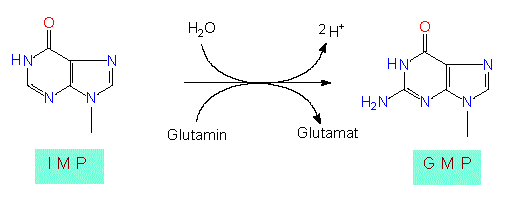
Depending on the structure of the base, it is distinguished between purines and pyrimidines. For the synthesis of a purine system, a molecule of aspartate, carbon dioxide, glycine and glutamine each and two formal-groups (transferred by tetrahydrofolic acid) are needed. For the initiation of synthesis, the already mentioned phosphoribosylphosphate is necessary. This means that already the first precursor of the base is coupled to a sugar and a phosphate residue. The first intermediate with a complete ring system is inosine monophosphate (IMP) the precursor of both AMP and GMP. The extensive list of starting compounds shows that the synthesis pathway is very energy-consuming. This is also the reason why purine residues are normally not degraded by the cell but are re-used and, if necessary, modified.
In the synthesis of pyrimidine, the ring-system is completed first. Starting compounds are aspartate and carbamyl phosphate. The latter is produced in the cytosol from the amino residue of the amino acid glutamine, free bicarbonate and the terminal phosphate residue of ATP:
glutamine + 2 ATP + hydrogen carbonate > carbamyl phosphate + 2 ADP + Pi + glutamate
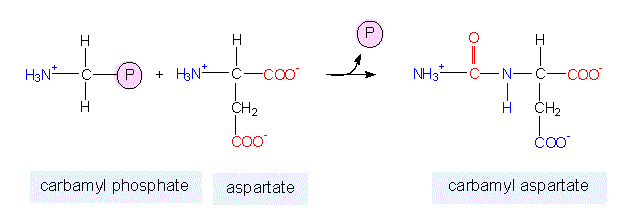
The heterocylic compound formed by the addition of aspartate is orotic acid that is now also bound to a phosphoribosylpyrophosphate. In this way, a nucleotide is produced. The splitting off of carbon dioxide (deletion of a side chain of the ring, a carboxy group) renders UMP that can be transformed into CMP by amidation.
© Peter v. Sengbusch - Impressum