
Botany online 1996-2004. No further update, only historical document of botanical science!
Plants are able to generate all 20 amino acids necessary for protein synthesis by themselves. They do even synthesize some more amino acids. We will meet several of them in the following sections.
The molecules we got to know when discussing glycolysis and the
citric acid cycle were carbon-containing but nitrogen-free. Some of
these intermediates are starting points of amino acid biosynthetic
pathways. Based on chemical similarities and only few starting
compounds, all amino acids can be regarded as members of five
families:
- the glutamate family starting with alpha-ketoglutarate
- the aspartate family with the starting compound oxaloacetate
- the alanine-valine-leucine group (pyruvate)
- the serine-glycine group (3-phosphoglycerate)
- the family of aromatic amino acids (phosphoenolpyruvate and erythrose-4-phosphate, an intermediate in the pentose phosphate pathway).
And where does the nitrogen come
from? Plants take up nitrogen as nitrate, in smaller amounts also
as ammonium ions. A few species, mainly of the leguminosa family,
live in symbiosis with nitrogen-fixing bacteria that are able to
reduce atmospheric nitrogen. All other plants reduce nitrate
(NO3-).
In a first step, nitrate is reduced to nitrite (NO2-). The reducing agent is NADH + H+ that is gained during glycolysis, the respective enzyme nitrate reductase. This reaction occurs in the cytosol and couples glycolysis and nitrate reduction and has the advantage that the NAD+ necessary for the progress of glycolysis.
In the second step, nitrite is reduced to ammonium by nitrite reductase, an enzyme that is located in plastids. Each type of plastid has its own assimilatory nitrite reductase. In photosynthetic tissues, reduced ferredoxin functions as the electron donor that is produced in photosynthesis.
The reactions are part of an electron transport chain that involves both NADP and FAD. The exact coupling of the steps is still partly unknown. Free ammonium ions are toxic to plant cells and are rapidly incorporated into organic compounds. The most important quantitative way in green plants is the reductive amination of alpha-ketoglutarate to produce glutamate catalyzed by the NADP+-dependent glutamate dehydrogenase. Glutamate is accordingly the amino acid generated first. Before following this pathway further and getting to know the glutamate family, the alternative pathway has to be mentioned. Glutamate itself can bind a further ammonium ion to form glutamine, a second amino acid. This reaction takes place in chloroplasts, too. Both reactions are ATP-dependent.
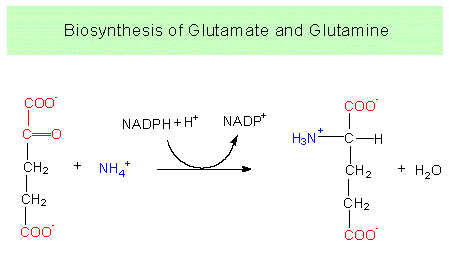
Glutamate dehydrogenase reaction: The starting compounds are alpha-ketoglutarate and NH4+.

Glutamate synthetase reaction: starting compounds are glutamate and NH4+.

Glutamate synthase reaction (a transamination). The amino group of an amino acid (glutamate) is transferred to an alpha-ketoacid (alpha-ketoglutarate).
Very important for amino acid syntheses is a group of enzymes
called aminotransferases. They are able to transfer an amino group
(mostly that of glutamine) to an alpha-keto acid. Accordingly,
they could also be said to be the distributors of amino groups.
Germinating seeds that have not started photosynthesis yet get their
amino groups exclusively by degradation of storage proteins. They are
subsequently fed into the outlined pathways to generate new amino
acids, proteins and other nitrogen-containing compounds.
The glutamate family. The amino acids glutamate, glutamine, proline and arginine are members of this family. The synthesis of the first two has just been discussed.
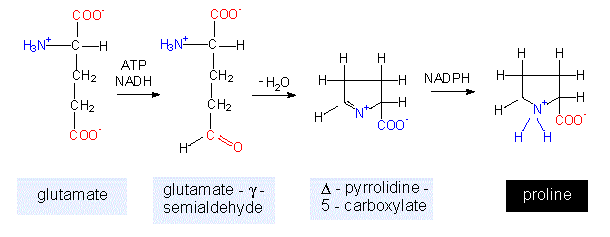
Proline is generated by a ring formation under consumption of one molecule of NADH + H+, NADPH + H+ and ATP each. This points out how energy-consuming biosyntheses are. Since it will not be mentioned explicitly in the following discussion, it should be kept in mind that every comparable biosynthetic pathway is fuelled by energy amounts of the same scale. Proline contains no primary but a secondary amino group and is therefore actually an alpha-imino acid (the name-giving feature is the >NH group), but it is nevertheless referred to as an amino acid. It is the one exception to the general structure of amino acids.
A conversion of proline to hydroxyproline occurs only at proline residues that are part of a polypeptide chain. Hydroxyproline is especially common in the cell wall protein extensin.
Arginine is an alkaline amino acid, i.e. it contains a second amino group that is ionized under physiological conditions. Its synthesis has eight intermediate steps. An important intermediate is ornithine that can be generated in plant cells by two alternative ways. It functions as an acceptor of carbamyl phosphate. At the same time, a phosphate is split off. The resulting next intermediate is citrulline.
Both intermediates belong to the group of amino acids that is common in cells but is not used in the synthesis of proteins. Binding of aspartate enlarges the molecule again. A part of the new intermediate is then split off (fumarate) generating arginine. This biosynthesis explains several points:
|
|
the biosynthesis of complex products involves a whole range of intermediates. |
|
|
a C-C chain is elongated by the coupling of two molecules. Accordingly, not every bond is formed anew for each product. |
|
|
amino acids are not only needed for the synthesis of proteins but also for that of other amino acids |
|
|
The aspartate family. The
amino acids apartate, asparagine, threonine, isoleucine and
methionine are counted among this group. Aspartate is generated by
the transamination
glutamate + oxaloacetate > alpha-ketoglutarate + aspartate
and asparagine by the addition of a further amino group. This may on one hand be achieved by the fixation of an ammonium ion (analogous to the synthesis of glutamine), on the other hand may it also occur by the transfer of one amino group from glutamine to aspartate.
NH4+ + aspartate + ATP > asparagine + ADP + Piglutamine + aspartate + ATP > glutamate + asparagine + AMP + PP
From aspartate branches a pathway to L-homoserine, an intermediate that is the starting point of the syntheses of both threonine and isoleucine, and methionine. The biosynthesis of threonine has again several intermediates and consumes two molecules of ATP, one NADPH + H+ and one NADH + H+. The energetically more favourable solution would be:
acetaldehyde + glycine > threonine
but acetaldehyde is in short supply in aerobic cells (apart from he fact that it is a powerful cytotoxin).
The synthesis of isoleucine starts with threonine, that of methionine needs sulphur. Sulphur is taken up by plants as sulphate ions and is subsequently converted to its reduced form in a two step reaction. The intermediate is sulphide. One enzyme catalyzing this reaction was isolated from spinach chloroplasts. It, too, needs the reduced ferredoxin as an electron donor. The reduced sulphur is finally converted to the -SH group of cysteine (assimilatory sulphate reduction).
It is then transferred to a precursor of methionine. On the other hand, a pathway by which cysteine can be regained from methionine exists, too. A cofactor in the methionine production is tetrahydrofolate (THF) that transfers a methyl group to the -SH group of homocysteine, the precursor of methionine. The result is the terminal -S-CH3 group of methionine.
Lysine synthesis. Intermediates of the glutamate and the aspartate family are coupled in just the same way as in the arginine and asparagine synthesis. Lysine can be generated in two ways. Both are named according to their most characteristic intermediates. Green algae, ferns and higher plants take one pathway, some green flagellates (euglenophyta) and fungi the other.
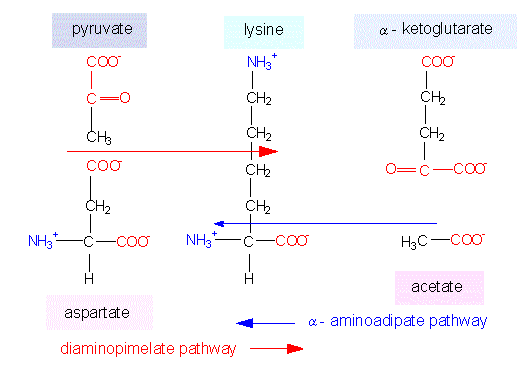
Two ways of Lysine Biosynthesis
The alanine-valine-leucine group has clearer conditions. Pyruvate is the starting compound, its amino derivative is alanine. Valine and leucine are synthesized independently of each other by elongation of the pyruvate chain, interconversion and subsequent transamination.
An additional remark: Though leucine and isoleucine have very similar structures and properties, they do have fundamentally different pathways.
The serine-glycine-cysteine
group. Serine is generated in a two-step reaction from
3-phosphoglycerate. The first step is an oxidation, the second a
transamination.

Alternatively, 3-phosphoglycerate can also be dephosphorylated to generate hydroxyproline that is subsequently converted to serine by transamination.
A path branches from serine to glycine in which the -CH2OH part of the serine side chain is split off and the C1 unit is accepted by the already mentioned tetrahydrofolate (THF). Another pathway results in cysteine. This means that cysteine can also be produced by at least two paths (the other was the degradation of methionine).
Aromatic amino acids need both phosphoenolpyruvate and erythrose-4-phosphate as starting compounds. A ring is formedvia several intermediates. Both shikimate and the subsequent chorismate are important intermediates. The latter is the starting compound for three different pathways that lead to the end products phenylalanine, tyrosine and tryptophane.
Several pathways for the synthesis of all these amino acids exist. An activated form of ribose phosphate (5-phosphoribosyl-alpha-pyrophosphate) is an intermediate of tryptophane synthesis. It has also a key position in the synthesis of histidine that is not discussed here and in that of purines and pyrimidines. It should only be mentioned that the biosynthetic pathway of histidine is unusual in that it is produced from a purine.
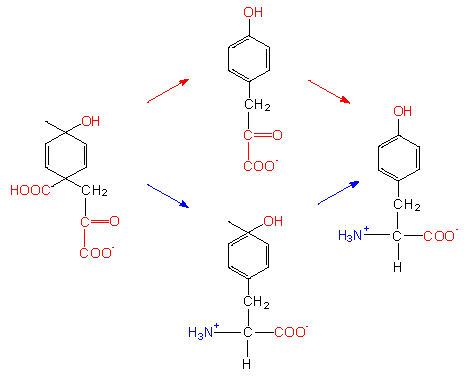
Alternative biosynthetic pathways. The starting compound shown in the picture to the left is an unstable intermediate that is generated from chorismate by the chorismate mutase. Chorismate is the aromatic amino acid precursor.