Botany online 1996-2004. No further update, only historical document of botanical science!


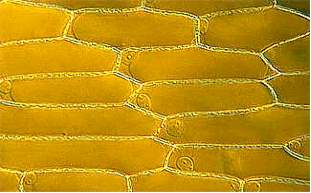
To depict the structure of a plant cell, an epidermal cell of an onion will be used as an example. The epidermis is the final tissue that covers all organs above ground. The cells of the onion epidermis are common specimens on the first day of a German basic botanical course. Since they contain no chlorophyll, they are actually no "typical" plant cells.
The picture above shows an onion"s epidermal cells. They are elongated and the ratio of length to width can vary strongly. Each cell is enclosed by a wall. In the region of the cell poles and where three cells adjoin, large intercellular spaces can be observed. Elsewhere a pectin-containing middle lamina cements neighboring cells together like bricks. The cell wall is perforated at regular intervalls, so that adjoining cells are in contact. The holes of the perforation are called simple pits and the plasma cords that run through them plasmodesmata . The surface of the epidermal cells seems to be folded, an effect that is caused by the water-repellent, waxy cuticle.
The "living" content of a cell, the protoplasm , is surrounded by a membrane called plasmamembrane or plasmalemma. The protoplasm is usually next to the cell wall, so that the plasmalemma can hardly be seen. To display it, the cells are transferred into a high salt or sugar solution. As a result the protoplasm shrinks and detaches itself from the wall. The process is reversible and is called plasmolysis. This behavior is due to the properties of the membrane and the plasma. It is reviewed in more detail elsewhere. A substance that causes plasmolysis is called plasmolyticum and - depending on its chemical composition (potassium ions or calcium ions, for example) - the protoplasm takes on different shapes. The plasmolyticum has accordingly an influence on the properties of the membrane. The properties of the plasmamembrane differ from that of the tonoplast. The tonoplast is the membrane that surrounds the vacuole. The difference is especially striking if cells with a colored vacuole content are used. Often the vacuole is criss-crossed by numerous plasma cords. The plasma cannot therefore not simply be viewed as a solution that is influenced by the rules of hydrodynamics alone. Rather, it contains viscous, structure-determining components, whose chemical, physicochemical and structural properties have only been recognized recently and in fragments.
The nucleus is a rather conspicuous part of nearly every living plant cell. Its structure separated from the rest of the cell by the nuclear envelope, a membrane system that consists of two discrete membranes as can be seen on electromicroscopic images. The nuclear content is called the caryoplasm while that of the rest of the cell is called cytoplasm. But these terms are only valid at certain stages of a cell's life cycle. In the course of cell-division and mitosis, the nuclear envelope disintegrates and the nucleus is replaced by the chromosomes. It makes consequently no sense to speak of caryo- and cytoplasm during these stages.
The nucleus of plant cells is usually of a round or elliptic appearance, sometimes it is also shaped like a spindle. One nucleus per cell is the rule, but cells with two or more nuclei are no rare exception. The cells of certain algae of the genus Chladophora have many nuclei, they are polyenergid. The nucleoli that can often be perceived after staining are substructures of the nucleus. They, too, disintegrate during cell division and mitosis and do not reshape before a new nucleus has been formed.
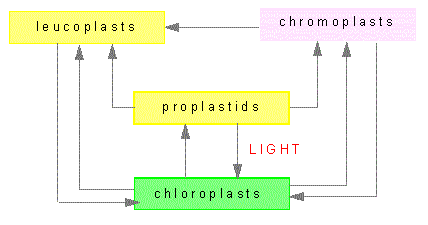
Plastids are organelles that occur only in plants. Their most prominent members are the chloroplasts. Others plastids are the colored chromoplasts and the colorless leucoplasts as well as their proplastids. Proplastids are vestigial bodies that are generated during germ cell development due to degeneration of plastids, for example. They may differentiate into complete plastids during the development of the plant embryo. Their ripening into chloroplasts occurs usually only after light exposure.
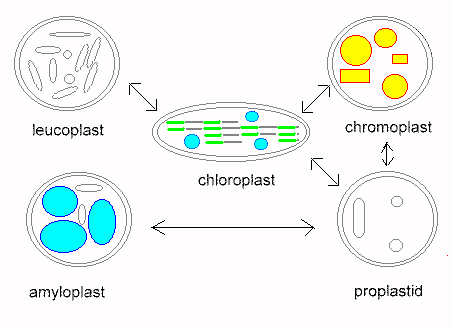
Chloroplasts contain the green plant color chlorophyll. They are the places where photosynthesis takes place. Chloroplasts enable the plants to convert solar energy into chemical energy. Because of this process, plants are called primary producers. The existence of consumers, like most animals, depends on them. Chloroplasts occur in most cell types, but only in organs above ground. They can be especially well observed in tissues consisting of a single layer as in the leaflike structures of some mosses (Funaria hygrometrica or Mnium hornum) or in the water plant Vallisneria). Here they are rather large and of a lens-shaped appearance. During daytime, when the light is diffuse, they occur mainly at the upper and lower surface of the chloroplast. They appear to be round under top view. If exposed to strong light, they gather in parallel to the lateral sides of the cell which gives them an elleptic appearance upon top view.
Chloroplasts are the site of starch production and -storage. Starch can easily be detected with the aid of potassium iodide (LUGOL's reagent). The starch-iodine complex is deeply blue-violet.
Starch production during photosynthesis can be made visible by placing a mask at a leaf that covers it partially while leaving some places exposed to sunlight. After one day of exposure, the leaf is first bleached to get rid of other pigments and afterwards treated with potassium iodide. An image is gained that is the exact replication of the mask and at the same time represents starch synthesis in the leaf. This experiment has first been done by J. v SACHS, probably the most outstanding plant physiologist of the 20th century. He thought that starch was the primary product of photosynthesis. This assumption proved wrong. It is well-known today that the first products of photosynthesis are monomeric sugars (glucose and others) and that only part of them is used for starch production.
The structures of the chloroplasts of higher cells resemble largely that of mosses. Their average diameter is 4 - 8 mm, an average cells contains 10 through 50. Their chlorophyll is unevenly distributed. At high resolutions chlorophyll-rich and chlorophyll-poor areas can be distinguished. This is due to the inner structure of the chloroplast: it is organized into grana (chlorophyll-rich) and stroma (chlorophyll-poor). Upon stimulation with short-waved light (blue or violet) the chlorophyll emits an intensive red autofluorescence that looks especially impressive in a fluorescence microscope. The differences between grana and stroma become very obvious. The uniformity of the chloroplasts of all higher plants points out that the optimal form has been found rather early in evolution and has not been changed since. This is different with algae. The chloroplasts of green algae (Chlorophyceens) are very varied in shape. Many species have just one chloroplast that covers nearly the whole space of the cell's interior. It is screw-like in Spirogyra-species, star-shaped in Zygnema and Zygnemopsis and netlike in Oedogonium.
The disc-shaped chloroplast of Mougeotia can be viewed either from above or in profile depending on the amount of light used. Its rotation is a well-analyzed example of an induced chloroplast movement. The chloroplasts of many species of algae contain often well-visible pyrenoids, structures, that produce and structure starch.
Chromoplasts are red, orange or yellow plastids. The color is usually the result of yellow xantophyll and yellow to red carotinoids. Both compounds do also exist in chloroplasts, but are concealed by chlorophyll. Chlorophyll is broken down much faster than carotinoids as can be observed in the colored leaves in autumn. Fluid transitions between chromo- and chloroplasts exist, just as between chromo- and leucoplasts. Typical chromoplasts cause the orange color of the carrot, the red color of the ripe pimento and tomato as well as the color of numerous flowers. Carotinoids are not very water-soluble and do therefore often crystallize within the chromoplasts. Their crystals can be disc-shaped, needle-like, jagged or sickle-like.
In many cases, flower and leaf colors are caused by the colored content of the vacuole. The color of the vacuole and that of the plastids may lead to a mixed color. The leaves of the copper beech, where the vacuole's content is red and that of the chloroplasts is green are a typical example. The plastids of the red and brown algae are traditionally counted among the chromoplasts although they contain chlorophyll. The green colour is concealed by the red phycoerythrin (Rhodophyceae) or the brown fucoxanthin (Phaeophyta).
Leucoplasts are common, colorless plastids. They develop from proplastids, but form no homogeneous group of their own. A part of them can differentiate into chloroplasts or chromoplasts at light exposure, while this is not due for others. The guard cells, for example, contain leucoplasts, that are permanently exposed to light without developing into chloroplasts. Leucoplasts do also occur within colorless leaves (variegated leaves) or plant parts. There exists a number of examples which show that they developed from chloroplasts that lost their ability to produce chlorophyll. There are even species, like Neottia, an orchid that cannot produce chlorophyll at all and are thus dependent on a parasitic or saprophytic lifestyle (saprophy is the feeding from dead organic material).
A second class of leucoplasts occurs within the non-green tissues of otherwise green plants. It is especially common within roots. Though these leucoplasts are capable to become green, they do usually not since they are not exposed to light. The leucoplasts of the calyptra (a calyptra is any hood or cap of cells protecting a plant part) contain starch and are therefore counted among the amyloplasts (starch-containing leucoplasts). They have, as is explained later, the function of statolithes, that have an important part in the perception of gravity (geotropism).
We got to know starch in the section above as a content of chloroplasts and leucoplasts (amyloplasts). It is produced by the polymerization of glucose residues, which again are products of photosynthesis. Since the plant is able to transport sugars from leaf to root or from leaf to seed and fruit, starch production can also take place in these organs. Different species produce starch grains of different shape. Since the shape of starch grains informs about their origin, they are helpful in the identification of seeds and other starch-containing plant parts. The following numbers show the variations in their diameters. Starch grains from potato tubers have a diameter of 70 - 100 µm, that of the endosperm of wheat 30 - 45 µm and that of corn endosperm 12 - 18 µm. Their shape reflects their development. The starch molecules are long-stretched and only sparsely branched. They are deposited within the plastids and their development begins at a so-called formation center from where it proceeds radially. Layer follows layer and the thickness of one layer is dependent on the average molecular length. A starch grain is therefore organized like a crystal (semi-crystalline). This can be shown very impressively with a polarization microscope. Within the moistened specimen can a layering be viewed that is dependent on the water content of the single molecular parts.
 A.
Model of a starch grain structure. The single lines symbolize starch
molecules. They are arranged in a radial pattern.
B. Layering of the starch grains.
a. Formation center and layer borders, b. Diagram of the refraction
conditions. The ordinate shows the refraction index. (according to A.
FREY-WYSSLING, 1938)
A.
Model of a starch grain structure. The single lines symbolize starch
molecules. They are arranged in a radial pattern.
B. Layering of the starch grains.
a. Formation center and layer borders, b. Diagram of the refraction
conditions. The ordinate shows the refraction index. (according to A.
FREY-WYSSLING, 1938)
The denser the molecular packaging, the less water is deposited. Layers with less water content refract light stronger than those with much water. After drying of the preparation no layering can be perceived any more. Depending the central or peripheral placing of the formation center, starch grains with either concentric or eccentric layering develop. The starch grains of graminaceous plants (wheat, corn, etc.) are usually concentric, while those of the potato are always eccentric.
Sometimes plastids with two to three formation centres occur in graminaceous plants. This leads to the development of several starch grains. During growth common outer layers may be formed.
Compound starch grains are typical for oat, they are built from a large number of smaller grains.
Starch grains in bean seeds (Phaseolus vulgaris) are very big, their shape is round or oval, the spacing of the layers is very regular. Their centers can easily be hollowed out by addition of water displaying radial ruptures in microscopic images. In the sap of Euphorbia splendens dumb-bell shaped starch grains can be found.

Many plant cells contain crystalline inclusions of different chemical composition and shape. Crystalline aggregations are called druses, bundles of needle-shaped crystals are termed raphids. Scanning electron microscopic images: Top picture: Calcium oxalate druse in the mesophyll cells of an oleander leaf (Nerium oleander). Typical druse shape of dicots. Middle picture: Calcium oxalate needles (raphids) of a vanilla root (Orchidaceae). Typical raphid bundle of monocots. Lowest picture: Silicate bodies of silicate cells in the epidermis of Schizachyrium sanguineum (a gramineaen species of the old world tropics). Characteristic mineralization of a gramineaen cell. [W. BARTHLOTT, MARTENS, 1979 (c), W. BARTHLOTT, unpublished (a, b)]
Except for very few examples, plant cells are surrounded by a cellulose containing cell wall. It is flexible and distortable during growth, but loses its ability for distortion after growth has stopped, while a limited flexibility remains. Because of these changes, it is distinguished between primary and secondary cell walls. As we will see when talking about electron microscopic pictures of the cell wall, both forms differ mainly in the arrangement of their cellulose microfibrils. While they are unorganized within the amorphous matrix of the primary cell wall, they are organized into several ordered layers that are arranged one on top of the other at right angles in the secondary cell wall. The secondary cell walls of many cells, especially those of vascular tissues, are incrusted with strengthening material. Two important ones are:
![]() lignin, the ground
substance of wood and
lignin, the ground
substance of wood and
![]() suberin, the ground
substance of cork
suberin, the ground
substance of cork
Their details are reviewed here. In addition, secondary walls contain often phenolic oxidation products that lends them a dark color (red to black with various shades).